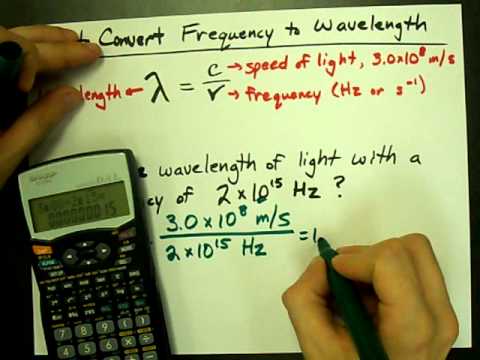SOLVED: Give reasons to convert the following spectroscopic quantities as indicated: (i) 0.15 nm to Hz (ii) 9 GHz to cm (iii) 2000 cm^-1 to km (iv) 500 nm to cm State

SOLVED: Give reasons to convert the following spectroscopic quantities as indicated: (i) 0.15 nm to Hz (ii) 9 GHz to cm (iii) 2000 cm^-1 to km (iv) 500 nm to cm State
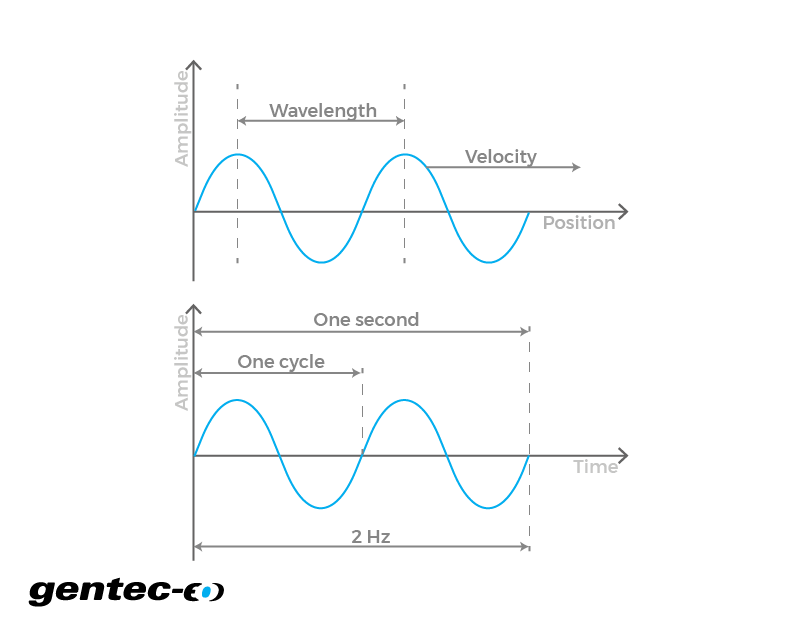
![EXAMPLE 1] The wavelength range of the visible spectrum extends from violet (400 nm) to red (750 nm). Express these wavelengths in frequencies (Hz). (1 nm = 10ºm) [NCERT Intext] Sol Given, EXAMPLE 1] The wavelength range of the visible spectrum extends from violet (400 nm) to red (750 nm). Express these wavelengths in frequencies (Hz). (1 nm = 10ºm) [NCERT Intext] Sol Given,](https://toppr-doubts-media.s3.amazonaws.com/images/5140776/cb7119db-3705-4113-a61a-14799a32df6d.jpg)